Understanding the Single-Use Bioprocessing Market
What Is Single-Use Bioprocessing?
The single-use bioprocessing market has become one of the fastest-growing segments within the biotechnology and biopharmaceutical industries. At its core, single-use bioprocessing refers to manufacturing systems that use disposable components instead of traditional stainless-steel equipment. These disposable components include bioreactors, tubing, bags, filters, and connectors that are used once and then discarded after production cycles. This approach significantly reduces cleaning and sterilization requirements, which are mandatory in conventional bioprocessing systems.
In biopharmaceutical production, maintaining sterile conditions is absolutely essential. Traditional stainless-steel systems require complex cleaning-in-place (CIP) and sterilization-in-place (SIP) procedures that can take hours or even days. Single-use technologies eliminate these steps entirely, offering a streamlined manufacturing process. The technology has proven particularly useful in the production of vaccines, monoclonal antibodies, recombinant proteins, and cell and gene therapies. As the biotechnology sector continues to evolve toward personalized medicine and small-batch biologics production, single-use systems provide the flexibility and scalability manufacturers require.
Biopharmaceutical companies and contract manufacturing organizations are increasingly integrating these disposable technologies to improve operational efficiency. The reduced infrastructure cost, lower contamination risk, and shorter turnaround times make single-use systems extremely attractive for both large-scale production and research environments. This paradigm shift is gradually transforming how biologics are produced across the global pharmaceutical industry.
Why Disposable Technologies Are Transforming Biomanufacturing
Imagine running a pharmaceutical factory where each batch requires hours of cleaning, sterilization, and validation before the next production cycle begins. That has been the reality of traditional biomanufacturing for decades. Disposable systems completely change that equation. By replacing reusable stainless-steel vessels with pre-sterilized, single-use equipment, manufacturers can begin a new production run almost immediately after finishing the previous one.
The biggest advantage of this model is flexibility. Biopharmaceutical companies often produce multiple biologic drugs using the same manufacturing facility. With conventional equipment, switching production lines requires lengthy cleaning procedures to prevent contamination. Single-use technology eliminates this issue, enabling faster product changeovers. This capability is particularly valuable in modern medicine, where therapies such as cell and gene therapy require small-scale but highly specialized production processes.
Another reason for the rapid adoption of disposable bioprocessing is cost efficiency. Building a traditional biomanufacturing facility with stainless-steel infrastructure can cost hundreds of millions of dollars. Single-use facilities require far less capital investment and significantly lower operational costs. This makes them especially attractive for emerging biotech companies and contract development and manufacturing organizations (CDMOs) that want to scale production quickly without massive infrastructure investments.
From improved sterility to faster production cycles, disposable technologies are redefining the economics and efficiency of pharmaceutical manufacturing. The shift toward flexible manufacturing systems is a strong indicator that single-use technologies will continue to dominate the future of biologics production.
Download PDF Broucher: https://www.marketsandmarkets.com/pdfdownloadNew.asp?id=231651297
Global Market Size and Forecast (2025-2030)
Market Valuation and CAGR Overview
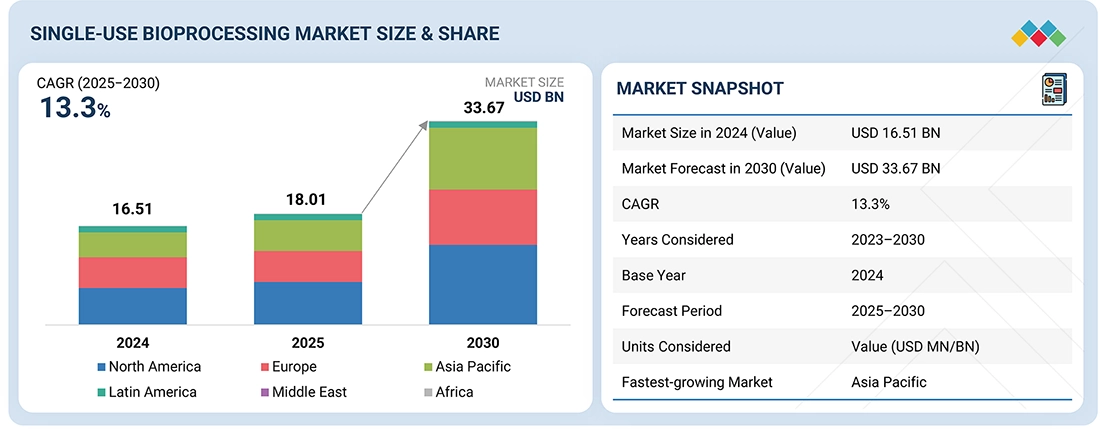
The global single-use bioprocessing market is experiencing remarkable growth as biotechnology companies increasingly adopt disposable manufacturing technologies. According to industry analyses, the market is projected to grow from USD 18.01 billion in 2025 to USD 33.67 billion by 2030, representing a compound annual growth rate (CAGR) of 13.3% during the forecast period.
This impressive growth trajectory reflects the expanding demand for biologics, vaccines, and advanced therapies. Over the past decade, pharmaceutical innovation has shifted away from traditional small-molecule drugs toward complex biological therapeutics. These therapies require specialized production environments where contamination risks must be minimized and manufacturing processes must remain highly flexible. Single-use systems address these challenges effectively, making them an essential component of modern biopharmaceutical facilities.
Another factor contributing to this market expansion is the rapid growth of contract manufacturing organizations (CMOs) and contract development and manufacturing organizations (CDMOs). These organizations provide outsourced production services for biotech companies that lack large manufacturing infrastructures. Disposable bioprocessing technologies enable CDMOs to quickly adapt their facilities for different clients and products, increasing operational efficiency while reducing downtime.
Additionally, global healthcare demand continues to rise due to aging populations, increased chronic disease prevalence, and emerging infectious diseases. Governments and pharmaceutical companies are investing heavily in vaccine production capabilities and biologics manufacturing capacity. This ongoing investment in advanced therapeutic development is directly driving the adoption of single-use technologies worldwide.
Key Market Statistics
The single-use bioprocessing market is characterized by strong growth indicators and widespread adoption across the biotechnology sector. Key statistics help illustrate the scale and momentum of this industry:
| Market Metric | Value |
|---|---|
| Market Size (2025) | USD 18.01 Billion |
| Forecast Market Size (2030) | USD 33.67 Billion |
| CAGR (2025-2030) | 13.3% |
| Leading Region | North America |
| Fastest Growing Region | Asia-Pacific |
These figures demonstrate how rapidly the industry is expanding. North America currently dominates the market due to its advanced biotechnology infrastructure and strong pharmaceutical presence. At the same time, Asia-Pacific is emerging as a key growth hub as countries such as China, India, and South Korea expand their biologics manufacturing capabilities.
Major Drivers Fueling Market Growth
Rising Demand for Biologics and Biosimilars
One of the strongest drivers behind the single-use bioprocessing market is the rapidly growing demand for biologic medicines. Biologics include therapies such as monoclonal antibodies, vaccines, recombinant proteins, and gene therapies. These treatments have revolutionized medicine by offering targeted therapies for diseases like cancer, autoimmune disorders, and rare genetic conditions.
Unlike conventional pharmaceutical drugs, biologics are produced using living cells and require complex manufacturing environments. Maintaining sterility and process control is essential for ensuring product safety and efficacy. Disposable bioprocessing technologies are particularly well-suited for this type of production because they reduce contamination risks and allow flexible manufacturing setups.
The rise of biosimilars is also contributing to market growth. Biosimilars are essentially lower-cost versions of existing biologic drugs that have lost patent protection. Many pharmaceutical companies are investing in biosimilar production to meet global demand for affordable biologic therapies. Single-use systems help manufacturers scale biosimilar production quickly while minimizing infrastructure costs.
Reduced Risk of Cross-Contamination
Another major factor driving adoption is the reduced risk of cross-contamination during pharmaceutical manufacturing. In traditional facilities, even the smallest residue from a previous production batch can contaminate the next one. This risk is especially critical when producing highly sensitive biologic therapies.
Single-use systems eliminate this concern because each production run uses a completely new set of sterile components. After the batch is completed, the disposable equipment is discarded, ensuring that the next process starts with a clean environment. This not only improves product quality but also simplifies regulatory compliance for pharmaceutical manufacturers.
Additionally, disposable systems significantly reduce the time required for equipment validation and cleaning procedures. Manufacturers can switch production lines much faster, which increases facility productivity and accelerates time-to-market for new therapies.
Core Components of Single-Use Bioprocessing Systems
Consumables
Consumables represent the largest product segment within the single-use bioprocessing market. These disposable components are essential for maintaining sterile environments throughout the production process. Consumables typically include filters, tubing, connectors, bags, and sampling systems used during biomanufacturing operations.
These components are designed to be pre-sterilized and ready for immediate use. Because they are discarded after a single production cycle, they eliminate the need for cleaning and sterilization procedures. This greatly reduces downtime and operational complexity in biopharmaceutical facilities.
Consumables play a critical role in maintaining sterility and preventing contamination during sensitive processes such as cell culture and purification. Their growing demand reflects the increasing adoption of disposable technologies across research laboratories, pilot plants, and large-scale commercial production facilities.
Equipment
In addition to consumables, the market also includes disposable equipment such as single-use bioreactors, filtration systems, and mixing systems. These technologies allow manufacturers to conduct complex biological processes using disposable containers and components.
Single-use bioreactors are particularly important because they serve as the primary vessel for cultivating living cells during biologic drug production. Modern single-use bioreactors can support volumes ranging from a few liters to several thousand liters, making them suitable for both clinical trials and commercial manufacturing.
Advancements in sensor technology and process automation are also enhancing the performance of disposable equipment. Integrated sensors can monitor parameters such as temperature, pH, oxygen levels, and pressure, ensuring precise control over bioprocessing conditions.
Applications Across Biopharmaceutical Manufacturing
Upstream Processing
Upstream processing involves the initial stages of biologics production, including cell line development, cell culture, and fermentation. This stage is responsible for producing the biological molecules that will eventually become therapeutic drugs.
Single-use technologies are particularly valuable during upstream processing because they provide flexibility and scalability. Researchers can quickly adjust production parameters and scale up processes without needing extensive infrastructure modifications. This capability is especially useful for early-stage drug development and clinical trials.
Downstream Processing
Downstream processing focuses on purification and recovery of biological products after they have been produced by cells. This stage typically includes filtration, chromatography, and concentration steps to ensure that the final product meets strict quality standards.
Disposable filtration systems and purification components help streamline downstream processing by reducing contamination risks and simplifying workflow integration. These technologies also allow manufacturers to maintain consistent product quality while increasing production efficiency.
Regional Market Analysis
North America Market Leadership
North America currently holds the largest share of the global single-use bioprocessing market. The region benefits from a strong biotechnology ecosystem, advanced research institutions, and a high concentration of pharmaceutical companies. In fact, North America accounted for a significant portion of global revenue in recent years due to widespread adoption of advanced biomanufacturing technologies.
Government support for biotechnology innovation, combined with substantial investment in biologics manufacturing infrastructure, continues to drive regional growth.
Asia-Pacific: The Fastest-Growing Region
Asia-Pacific is expected to experience the fastest growth in the coming years. Countries such as China, India, South Korea, and Singapore are rapidly expanding their biotechnology industries. Governments in these regions are investing heavily in pharmaceutical manufacturing capacity to reduce dependence on imported biologics.
The lower operational costs and growing talent pool in Asia-Pacific also make it an attractive location for contract manufacturing organizations.
Competitive Landscape
Key Companies in the Market
The single-use bioprocessing industry is highly competitive, with several major biotechnology equipment manufacturers dominating the market.
| Key Company | Industry Role |
|---|---|
| Thermo Fisher Scientific | Bioprocessing equipment & reagents |
| Sartorius AG | Bioprocessing solutions |
| Danaher Corporation | Biotechnology instrumentation |
| Merck KGaA | Life science technologies |
| Avantor | Bioprocessing materials |
| Corning Incorporated | Cell culture systems |
These companies focus heavily on innovation, product development, and strategic partnerships to expand their market presence.
Challenges and Limitations
Despite its rapid growth, the single-use bioprocessing market faces several challenges. One of the primary concerns is waste management. Because disposable systems generate plastic waste after each production cycle, environmental sustainability remains a key issue.
Another challenge is the lack of standardized components across different manufacturers. Variations in equipment design can create compatibility issues within integrated manufacturing systems. Regulatory compliance also remains a complex process, especially when introducing new disposable technologies into pharmaceutical production environments.
Emerging Trends and Future Opportunities
Looking ahead, several emerging trends are shaping the future of single-use bioprocessing. One of the most exciting developments is the integration of automation and artificial intelligence into biomanufacturing processes. AI-driven analytics can monitor production parameters in real time, optimizing efficiency and improving product consistency.
Another major trend is the growing demand for cell and gene therapies, which require flexible manufacturing platforms capable of producing small, personalized batches of biologic medicines. Single-use technologies are ideally suited for these applications because they allow rapid process adaptation.
Sustainability is also becoming a priority. Many companies are developing recyclable materials and eco-friendly disposable systems to reduce environmental impact while maintaining production efficiency.
Conclusion
The single-use bioprocessing market is rapidly transforming the global biopharmaceutical manufacturing landscape. With the market projected to grow from USD 18.01 billion in 2025 to USD 33.67 billion by 2030, disposable technologies are becoming a cornerstone of modern biologics production.
The shift toward flexible manufacturing, increasing demand for biologics, and the expansion of CDMOs are all driving the adoption of single-use systems. While challenges such as waste management and standardization remain, ongoing technological innovation is expected to address these issues. As biotechnology continues to evolve, single-use bioprocessing will play a crucial role in enabling faster, safer, and more efficient production of life-saving therapies.
FAQs
1. What is the single-use bioprocessing market?
The single-use bioprocessing market refers to technologies and equipment used in biopharmaceutical manufacturing that rely on disposable components instead of reusable stainless-steel systems.
2. Why are single-use systems important in biomanufacturing?
They reduce contamination risk, lower infrastructure costs, and allow faster production cycles compared to traditional manufacturing equipment.
3. Which industries use single-use bioprocessing technologies?
Biopharmaceutical companies, biotechnology firms, contract manufacturing organizations, and research institutes commonly use these technologies.
4. What are the main components of single-use bioprocessing systems?
Key components include single-use bioreactors, filtration systems, tubing, bags, connectors, and sensors.
5. Which region leads the single-use bioprocessing market?
North America currently leads the market due to strong biotechnology infrastructure and high investment in biologics manufacturing.